|
3/29/2024 0 Comments Bohr atomic theory Under the Bohr model of the atom, what energy level is the electron in? Use a value of 1. Note that the unit J⋅s is equivalent to kg⋅m 2⋅s −1, which is commonly used for angular momentum.Įxample 2: Calculating the Energy Level of an Electron from Its Angular MomentumĪn electron in a hydrogen atom has an angular momentum of 3. Now that we have established that the electron in this question has □ = 1, we can just substitute this into the above equation to find the angular momentum: The fact that this is a hydrogen atom is relevant because the Bohr model is generally only accurate for single-electron atoms, and hydrogen atoms have only one electron. We can recall that the ground state of an atom is the lowest energy level, and that the principal quantum number of an electron in the ground state will take the lowest possible value, □ = 1. In this question, we are looking for an electron in the ground state of a hydrogen atom. Where □ is a positive integer known as the principal quantum number. Specifically, it tells us that □ can only be an integer multiple of the reduced Planck constant ℏ. The Bohr model of the atom tells us that the angular momentum □ of an electron in an atom is quantized. 0 5 × 1 0 J⋅s for the reduced Planck constant. In the Bohr model of the atom, what is the magnitude of the angular momentum of an electron in a hydrogen atom in the ground state? Use a value of 1. We just need to take the electron’s principal quantum number (which denotes its energy level) and multiply this by ℏ.Įxample 1: Calculating the Angular Momentum of an Electron in a Hydrogen Atom The equation □ = □ ℏ makes it easy to calculate the angular momentum of an electron in an atom. It is worth noting that the unit of ℏ can also be expressed as kg⋅m 2⋅s −1, which is the usual unit we would use to represent angular momentum. The “reduced” Planck constant is equal to the ordinary Planck constant, ℎ, divided by 2 □,Īnd it has a value of 1. The principal quantum number denotes the energy level of an electron, where □ = 1 corresponds to the lowest possible energy state (also known as the ground state). □ is just some positive integer, but we give this a special name: the principal quantum number. Where □ is the angular momentum of the electron and ℏ (pronounced “h bar”) is the reduced Planck constant. Specifically, the Bohr model tells us that the angular momentum of an electron in an atom must be equal to a multiple of a constant known as the reduced Planck constant, ℏ. In addition to this, the Bohr model goes one step further: it states that the angular momentum of an orbiting electron is quantized, meaning it can only take specific values. The cubic model, proposed in 1902, described atoms as cubes of positively charged matter with negatively charged electrons at the vertices.Note that these diagrams are not to scale. Historically, scientists have used a variety of models to describe atoms. If an experiment reveals that a model is inaccurate, then physicists need to either change the model or develop a new one that can accurately describe the observed experimental results. 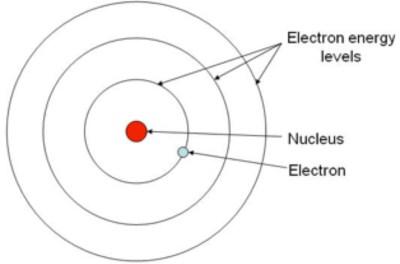
A model can be put to the test by experiments, which compare the predictions of the model to the observed behavior of a physical system. Physicists use models to describe how physical systems behave. 
In this explainer, we will look at the Bohr model in detail and see how we can use it to accurately calculate the angular momentum and orbital radius of an electron in an atom. It states-among other things-that electrons in atoms occupy circular orbits around the nucleus, similar to the way planets orbit the sun. 
The Bohr model is a simplified description of the atom. For example, copper and chlorine can form a green, crystalline solid with a mass ratio of 0.558 g chlorine to 1 g copper, as well as a brown crystalline solid with a mass ratio of 1.116 g chlorine to 1 g copper.In this explainer, we will learn how to calculate the orbital radius of an electron in different energy levels of a hydrogen atom. The law of multiple proportions states that when two elements react to form more than one compound, a fixed mass of one element will react with masses of the other element in a ratio of small, whole numbers. For example, there are many compounds other than isooctane that also have a carbon-to-hydrogen mass ratio of 5.33:1.00.ĭalton also used data from Proust, as well as results from his own experiments, to formulate another interesting law. 
That is, samples that have the same mass ratio are not necessarily the same substance. It is worth noting that although all samples of a particular compound have the same mass ratio, the converse is not true in general. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
